|
If you took two ethanoic acid molecules and removed a molecule of water between them you would get the acid anhydride, ethanoic anhydride (old name: acetic anhydride). A carboxylic acid such as ethanoic acid has the structure: 16 Evidence for strong interaction of their acyl - and Boc-substituents with nitrogen was noticed in both molecules. It introduces their chemical reactivity in a general way. In order to study the resulting bonding geometry, the 3D structure of two Boc-compounds of this type, one containing acetyl and one benzoyl, was determined by X-ray crystallography. Figure S.2: 1H NMR spectra (300 MHz, CDCl3) of N-Boc -3-amino- 1-propanol. T his page explains what acid anhydrides are and looks at their simple physical properties such as boiling points. Synonyms Di-t-butyl-dicarbonate DIBOC Boc Anhydride Butyl dicarbonate, Di-tert- Di-tert-butyl. CH2), 3.08 (m, CH anhydride), 7.25 (br m, Ar CH). Acid Anhydrides react with amines to form amides.Acid Anhydrides react with alcohols to form esters.The Fmoc-protected PNA backbone is a key intermediate in the synthesis of nucleobase-modified PNA monomers. The synthesis was performed starting with 50 g of Boc anhydride to give 31 g of product in 32 overall yield. Uses advised against Food, drug, pesticide or biocidal product use. Acid Anhydrides react with water to form carboxylic acids The Boc group is removed and replaced with an Fmoc group. Synonyms BOC anhydride DIBOC Di-tert-butyl pyrocarbonate Recommended Use Laboratory chemicals.Care should be taken when running boc deprotections because it is a gas forming reaction.ġ) Patent Reference: WO2014177977, page 65, (6.\) BOC Sciences can provide a wide variety of raw materials from upstream lines for automotive parts manufacturing, such as monomers, polymers and additives. Since this compound can be regarded formally as the acid anhydride derived from a tert-butoxycarbonyl (Boc) group, it is commonly referred to as Boc anhydride. 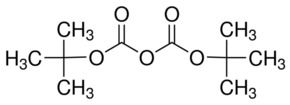
īoc anhydride causes eye and skin irritation and is harmful if inhaled. Synonym, di-tert-butyl dicarbonate, boc anhydride, boc-anhydride, di-t-butyl dicarbonate, di-tert-butyl pyrocarbonate, bis tert-butoxycarbonyl oxide, di-tert. 
To a solution of the SM (15 g, 43 mmol) and (Boc)2O (23 g, 105 mmol) in DCM (150 mL) was added TEA (19 mL, 140 mmol). Aldrich-205249 Di-tert-butyl dicarbonate ReagentPlus(R), 99 CAS No.: 2 Synonyms: Boc anhydride Di-tert-butyl pyrocarbonate Linear Formula. In order to use boc anhydride as a solid, it should be kept in a refrigerator and weighed out soon after being removed from the refrigerator. It often starts to melt while being weighed out for a reaction. The formation of Boc-protected amines and amino acids is conducted under either aqueous or anhydrous conditions, by reaction with a base and the anhydride. Boc anhydride has a melting point of only 22-24 C. Boc homolog (mouse), BOC and IDBG-50192 and ENSG0000014483, protein binding, Plasma membranes, Boc and IDBG-162156 and ENSMUSG00000022606, BOC and IDBG-632359 and ENSBTAG00000013918.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
